Your body's immune system is already pretty good at spotting troublemakers. But cancer cells? They're the con artists of biology - wearing disguises, forging IDs, and generally making themselves very hard to catch. Traditional chemotherapy's solution has been to carpet-bomb the whole neighborhood and hope the bad guys go down with it. Spoiler: the collateral damage is brutal. But what if we could build a GPS-guided missile instead?
That's basically what antibody-drug conjugates - ADCs for short - are. And according to a sweeping new review in Physiological Reviews by Cheung and colleagues, this field is absolutely exploding ([1]).
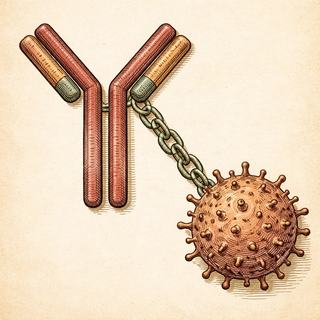
The Three-Part Weapon You Didn't Know Existed
Picture an ADC like this: you take a super-precise homing device (the antibody), strap a tiny bomb to it (the cytotoxic payload), and connect them with a molecular leash (the linker) that only snaps once it's inside the right cell. The antibody locks onto proteins that cancer cells wear like name tags on their surface. Once it docks, the whole package gets swallowed up by the cell, the linker breaks apart in the acidic mosh pit of the cell's lysosome, and the toxic payload goes to work - shredding DNA or jamming the cell's ability to divide.
The kicker? These payloads are often 100 to 1,000 times more potent than standard chemo drugs. You'd never dare put them into the bloodstream naked. But wrapped in an antibody's targeting embrace, they become precision instruments.
From Lab Curiosity to 430 Clinical Trials (and Counting)
Here's where the numbers get wild. Just a decade ago, only about 90 ADCs had entered clinical trials. Today? Over 430, with 14 earning FDA approval and more than 1,300 clinical trials actively running worldwide ([1], [2]). That's not growth - that's a stampede.
The poster child for this revolution is trastuzumab deruxtecan (brand name: Enhertu). It targets HER2, a protein found on many breast cancer cells. But here's the plot twist that shook oncology: Enhertu works even in cancers previously considered "HER2-low" - tumors with so little HER2 that older drugs couldn't touch them. In the landmark DESTINY-Breast04 trial, patients lived nearly twice as long without their cancer progressing compared to standard chemo ([3]). That single result redefined how we categorize breast cancer.
And Enhertu pulls off another trick called the "bystander effect" - once the drug payload is released inside one cancer cell, it can leak out and kill neighboring tumor cells too, even ones that don't have much HER2 on their surface. It's like taking out the target and their sketchy roommates.
The Engineering Arms Race
What makes the current generation of ADCs so much better than the clunky prototypes of the 2000s? Better everything, basically.
Smarter linkers that don't fall apart in the bloodstream but reliably snap inside tumors. Nastier payloads - topoisomerase I inhibitors (the engine behind Enhertu's success) have largely replaced the older tubulin inhibitors that earlier ADCs relied on. More precise conjugation chemistry that controls exactly how many drug molecules attach to each antibody - a number called the drug-to-antibody ratio, or DAR - because too few means weak; too many and the whole thing becomes unstable and toxic ([1]).
Researchers are also getting creative with targets. Beyond the usual suspects like HER2 and Trop-2, the review highlights emerging antigens like c-Met (which just earned an FDA approval via the ADC telisotuzumab vedotin for lung cancer) and Nectin-4 in bladder cancer.
Tag-Teaming with Immunotherapy
Perhaps the most exciting frontier is pairing ADCs with immune checkpoint inhibitors. It turns out ADCs don't just poison cancer cells - they can also shake up the tumor microenvironment in ways that make immunotherapy work better. By killing tumor cells in an immunogenic way, ADCs can increase CD8+ T-cell infiltration and reduce the immunosuppressive cells that tumors recruit as bodyguards ([2]). In urothelial cancer, the combination of the ADC enfortumab vedotin with pembrolizumab nearly doubled overall survival compared to chemo alone - from 16 months to over 31 months ([4]).
AI Enters the Chat
And because nothing in 2026 is complete without an AI angle: machine learning is now being used to predict optimal drug-to-antibody ratios, design better linkers, and identify new tumor targets from multi-omics data. Deep learning models like ADCNet are crunching antibody sequences and payload structures to predict which combinations will actually work before anyone touches a test tube ([5]). The era of trial-and-error ADC design is slowly giving way to something more like guided engineering.
What This Means for Actual Humans
The bottom line from this review: ADCs have evolved from a promising-but-finicky concept into one of the most dynamic areas of cancer drug development. With smarter designs, combination strategies, AI-assisted optimization, and an ever-growing list of targets, the next wave of ADCs could offer more personalized, more effective, and better-tolerated treatments than traditional chemo - or even current ADCs - can deliver.
Cancer cells have been dodging our best shots for decades. ADCs are learning to aim better.
References
-
Cheung A, Liu Y, Chenoweth AM, et al. Antibody-drug conjugate design and mechanisms of action for cancer treatment: state of the art and beyond. Physiological Reviews. 2026. DOI: 10.1152/physrev.00039.2025. PMID: 41855081.
-
Li Y, Wang Y, Zhang J, et al. Antibody-drug conjugates in cancer therapy: current landscape, challenges, and future directions. Molecular Cancer. 2025;24:163. DOI: 10.1186/s12943-025-02489-2. PMCID: PMC12581584.
-
Modi S, Jacot W, Yamashita T, et al. Trastuzumab deruxtecan in previously treated HER2-low advanced breast cancer. New England Journal of Medicine. 2022;387(1):9-20. DOI: 10.1056/NEJMoa2203690. PMID: 35665782.
-
Powles T, Valderrama BP, Gupta S, et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. New England Journal of Medicine. 2024;390(10):875-888. DOI: 10.1056/NEJMoa2312117. PMID: 38507751.
-
Tsuchikama K, Anami Y, Ha SYY, Yamazoe S. Exploring the next generation of antibody-drug conjugates. Nature Cancer. 2024;5:22-29. DOI: 10.1038/s43018-023-00707-2. PMID: 38191923.
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
Get cancer research delivered to your inbox
The best new studies, explained without the jargon. One email per week.