In real estate, they say it's all about location - and the same turns out to be true inside your body. Your pancreas, that unassuming little organ tucked behind your stomach, is normally a quiet residential district where hardworking acinar cells pump out digestive enzymes like clockwork. But when chronic pancreatitis moves in, the whole neighborhood goes downhill fast. And according to a striking new study published in Gut, if you've got the wrong genetic landlord, the block doesn't just deteriorate - it rolls out the red carpet for cancer.
The Worst Roommate Situation in Biology
Here's what researchers at Universitätsmedizin Halle figured out: they took a known troublemaker - a mutant version of the KRAS gene (basically the molecular equivalent of a con artist who's been implicated in about 90% of pancreatic cancers) - and paired it with a genetic mutation in CPA1, a digestive enzyme gene that causes hereditary chronic pancreatitis in humans.
In other words, they built a mouse that has both a pancreas perpetually inflamed AND a genetic predisposition to cancer. The resulting "KC-Cpa1" mouse model is, basically, the most realistic simulation we've ever had of what happens in people with hereditary pancreatitis who go on to develop pancreatic cancer (Inamdar et al., 2026).
And the results? Not subtle.
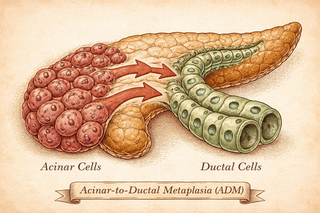
When Good Cells Go Bad (Like, Really Bad)
Normally, your pancreatic acinar cells are the reliable blue-collar workers of digestion - they make enzymes, they secrete enzymes, they go home. But under chronic inflammation, these cells undergo something called acinar-to-ductal metaplasia, or ADM. Basically, the factory workers start pretending to be office managers. They change shape, change function, and stop doing their actual jobs.
In the KC-Cpa1 mice, this identity crisis was dramatically worse. The CPA1 mutation causes the enzyme to misfold inside cells, triggering endoplasmic reticulum stress - imagine a factory where the assembly line keeps spitting out defective products that pile up until the building's fire alarm goes off. This stress signal doesn't just damage cells; it rewires them (Bhatt et al., 2018).
The researchers also discovered a brand-new type of troublemaker cell they're calling "iDucts" - inflammatory ductal cells that are basically the neighborhood gossips, broadcasting inflammatory signals to immune cells and fibroblasts, recruiting them into a tissue-remodeling frenzy. The result: more fibrosis, more metaplasia, more precancerous lesions called PanINs.
Why Hereditary Pancreatitis Is Playing With Fire
People with hereditary pancreatitis have a roughly 40% cumulative risk of pancreatic cancer by age 70 - that's about 50 times higher than the general population (Lowenfels et al., 1997). We've known this statistic for decades, but the why has remained frustratingly murky.
This study starts filling in that gap. The chronic inflammation from the CPA1 mutation doesn't just sit there being annoying - it actively cooperates with mutant KRAS. It's like inflammation kicks open the door and KRAS walks right through. The cellular plasticity created by pancreatitis gives cancer's favorite oncogene exactly the destabilized environment it needs to start building precancerous lesions (Kandikattu et al., 2020).
Single-cell RNA sequencing revealed that the crosstalk between ductal cells, immune cells, and fibroblasts creates disease-specific signaling networks - basically, the bad neighborhood develops its own communication system that helps it stay bad (Storz, 2017).
What This Actually Means for People
Pancreatic cancer is notoriously diagnosed late, when options are limited. This mouse model is the first to faithfully recapitulate hereditary pancreatitis-driven cancer from the very beginning, giving researchers a window into the earliest stages of the disease - stages we almost never get to see in human patients.
The discovery of those iDuct cells is particularly exciting. If these inflammatory ductal cells can be detected in humans, they could serve as early biomarkers - basically, a smoke alarm that goes off before the whole house burns down. And understanding the specific signaling networks involved opens the door to targeted interventions that could disrupt the inflammation-to-cancer pipeline before it reaches the point of no return.
For the roughly 1 in 10,000 people with hereditary pancreatitis, that's not just academic interest. That's hope with a molecular address.
References:
-
Inamdar TV, Krannich F, Hesselbarth N, et al. Hereditary chronic pancreatitis induced plasticity cooperates with mutant Kras in early pancreatic carcinogenesis. Gut. 2026;75(5):1003-1014. DOI: 10.1136/gutjnl-2025-335947. PMID: 41419303.
-
Lowenfels AB, Maisonneuve P, DiMagno EP, et al. Hereditary pancreatitis and the risk of pancreatic cancer. J Natl Cancer Inst. 1997;89(6):442-446. PMID: 9091646.
-
Bhatt B, Rösler S, Bhatt DK, et al. Human CPA1 mutation causes digestive enzyme misfolding and chronic pancreatitis in mice. Gut. 2019;68(2):301-312. PMID: 30045879.
-
Kandikattu HK, Venkateshaiah SU, Mishra A. Chronic pancreatitis and the development of pancreatic cancer. Endocr Metab Immune Disord Drug Targets. 2020;20(8):1182-1210. DOI: 10.2174/1871530320666200423095700. PMCID: PMC7577953.
-
Storz P. Pancreatic acinar-to-ductal metaplasia and pancreatic cancer. Methods Mol Biol. 2019;1882:299-317. DOI: 10.1007/978-1-4939-8879-2_26. PMID: 30378064.
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.