The cancer cells lost. Not just lost—they got outmaneuvered, outflanked, and then demolished by their own recycling machinery while simultaneously rusting from the inside out. That's the headline from a new study in Angewandte Chemie, where researchers essentially built a Trojan horse that sneaks in a protein-destroying weapon and then sets the whole fortress on fire.
The Problem: Prostate Cancer That Won't Quit
Castration-resistant prostate cancer (CRPC) is the chess grandmaster of malignancies. Standard treatments try to cut off testosterone—the fuel that prostate cancer cells crave—but CRPC has learned to play without that piece on the board. These tumors keep the androgen receptor (AR) signaling pathway running even when hormone levels drop to nothing, like a car that somehow keeps driving after you've drained the gas tank.
Enter PROTACs (Proteolysis-Targeting Chimeras), a class of drugs that don't just block proteins—they mark them for complete destruction by hijacking the cell's own protein disposal system. ARV-771 is one such PROTAC designed to tag AR proteins for demolition. Brilliant strategy on paper. One problem: getting ARV-771 into tumor cells without it wandering off to cause trouble elsewhere is like trying to deliver a pizza to a specific apartment in a building with no addresses.
The New Playbook: A Nanoparticle with Multiple Jobs
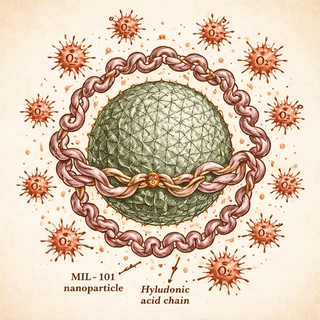
Wang and colleagues didn't just solve the delivery problem—they turned the delivery vehicle into a second weapon. Their creation, the delightfully named ARV@MIL-HA-ss-HA, is a nanoparticle system that would make military strategists nod approvingly.
Here's the tactical breakdown:
The carrier: MIL-101 nanoparticles serve as the transport vessel. But these aren't passive containers—they're nanozymes, meaning they have catalytic activity that converts hydrogen peroxide into hydroxyl radicals. Translation: once inside the cell, they start generating the chemical equivalent of tiny grenades.
The targeting system: The nanoparticles are coated with hyaluronic acid (HA), which homes in on CD44 receptors that CRPC cells overexpress. It's like the cancer cells put up a "deliver here" sign, and the nanoparticles are happy to oblige.
The release trigger: A disulfide bond (the "ss" in the name) links two HA molecules together. Inside cancer cells, elevated glutathione (GSH) levels break this bond, releasing the ARV-771 payload precisely where it's needed. Glutathione is the cell's main antioxidant defense, so depleting it serves double duty.
Checkmate: Two Killing Mechanisms Converge
The real genius is how these components work together to trigger ferroptosis—a type of cell death driven by iron-dependent lipid peroxidation. Normally, cancer cells have robust defenses against this kind of oxidative assault. But the ARV@MIL-HA-ss-HA system systematically dismantles those defenses:
- ARV-771 degrades AR and related proteins, disrupting multiple survival pathways
- The HA-ss-HA shell consumes intracellular GSH, stripping away antioxidant protection
- MIL-101's catalytic activity pumps out reactive oxygen species
- Without AR signaling or adequate antioxidants, the cancer cells become sitting ducks for ferroptosis
Previous research has established that combining protein degradation strategies with ferroptosis inducers can overcome therapeutic resistance in various cancers [PMCID: PMC9577974]. The convergence of these mechanisms creates what researchers call "synergistic sensitization"—each attack makes the cells more vulnerable to the other attacks.
Why This Matters Beyond the Lab
CRPC affects roughly 10-20% of prostate cancer patients, and treatment options remain frustratingly limited. Current therapies like enzalutamide and abiraterone eventually fail as resistance develops. The five-year survival rate for metastatic CRPC hovers around 30%, a statistic that hasn't budged much in years [PMCID: PMC8867685].
What makes this nanoplatform approach promising is its multi-pronged attack strategy. Drug resistance typically evolves when cancer cells find workarounds to single mechanisms of action. Attacking through protein degradation AND ferroptosis AND GSH depletion simultaneously gives cancer cells far fewer escape routes.
The improved pharmacokinetics are equally important. Free ARV-771 gets cleared from the body quickly and distributes non-specifically. The nanoparticle formulation extends circulation time and concentrates the drug at tumor sites, potentially meaning lower doses with better results and fewer side effects.
The Road Ahead
This is still preclinical work—the jump from promising lab results to approved therapy is long and littered with failures. But the fundamental strategy of using smart nanocarriers to deliver PROTACs while simultaneously sensitizing tumors to alternative cell death pathways represents a legitimate tactical innovation.
For now, CRPC patients remain locked in a difficult battle. But studies like this suggest reinforcements are being designed—ones that don't just fight cancer on one front, but coordinate a multi-directional assault that leaves resistant tumors with nowhere to run.
References:
-
Wang C, Jiang X, Lei J, et al. Nanozyme-Mediated PROTACs Delivery for Targeted Protein Degradation and Ferroptosis Sensitization in Prostate Cancer. Angew Chem Int Ed. 2025. DOI: 10.1002/anie.6657773
-
Zhang X, et al. Ferroptosis in cancer therapy: mechanisms, small molecule inducers, and novel approaches. Acta Pharm Sin B. 2021;11(8):2150-2166. PMCID: PMC9577974
-
Sumanasuriya S, De Bono J. Treatment of Advanced Prostate Cancer—A Review of Current Therapies and Future Promise. Cold Spring Harb Perspect Med. 2018;8(6):a030635. PMCID: PMC8867685
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.